 
So here we suppose another model as shown in Fig. 1., the two electrons are just at the opposite positions, so the wave phases of them may interfere with each other and vanish. 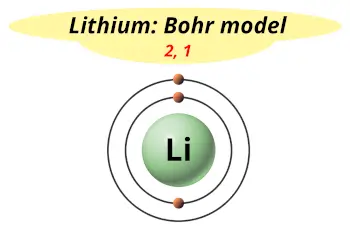
If the two electrons can be in one small orbit of one de Broglie's wavelength, this means that the ground state electron of the Bohr hydrogen-like model can come closer to the nucleus than the original orbit.Īnd in the orbit of Fig. This value is lower than the experimental value -198.09 eV. Solving the above three equations, the ground state energy (n=1) becomes -205.79 eV. The total energy E of the lithium ion (Li+) is the sum of the kinetic and the Coulomb potential energy of the two electrons, so Where h is Plank's constant (= 6.62606896 x 10 -34 Js), and h/mv is the de Broglie's wavelength. The circular orbital length is supposed to be an integer times the wavelength of the electron, we have Where r is the circular orbital radius (meter), m is the electron mass (me= 9.1093826 x 10 -31 kg), e is the electron charge (= 1.60217653 × 10 -19 C), and ε is the permittivity of vacuum (= 8.854187817 × 10 -12 C 2/Nm 2). One schematic model of lithum ion (Li+)Įquating the centrifugal force to the Coulomb force, we have 1.) in which two electrons of the lithium ion are on the opposite sides of the nucleus and moving on the same circular orbit.įig. Lithium ion (Li+) has two electrons and one nucleus (3e+).įirst, suppose we have one model (Fig. So the ground state energy of the lithium ion (Li+) is -75.64 - 122.45 = -198.09 eV. The ionization energies of the lithium is 5.39 eV (1st), 75.64 eV (2nd), and 122.45 eV (3rd), respectively. Naturally occurring lithium is composed of two stable isotopes, Li6 and Li7, the latter being the more abundant (92.5%). because of this, it is a good conductor of both heat and electricity, and used for the lithium (ion) batteries. Lithium has a single valence electron ( its configuration, 1S × 2, 2S × 1 ) that is easily given up to form a cation. It is the lightest metal, and highly reactive and flammable (though more stable than the other alkali metals). Lithium belongs to the alkali metal group of chemical elements, and has the atomic number 3. So next, we try Lithium atom (Li) and Lithium ion (Li+) by Bohr's theory. Our new Bohr model has suceeded in calculating the Helium ionization energy more correctly than the quantum mechanical variational methods as shown in the Top page. Top page (correct Bohr model including the two-electron atoms). Thus, the columns of the periodic table represent the potential shared state of these elements' outer electron shells that is responsible for their similar chemical characteristics.Bohr's Lithium(ion) Li(+) New Bohr model Lithium (Li) 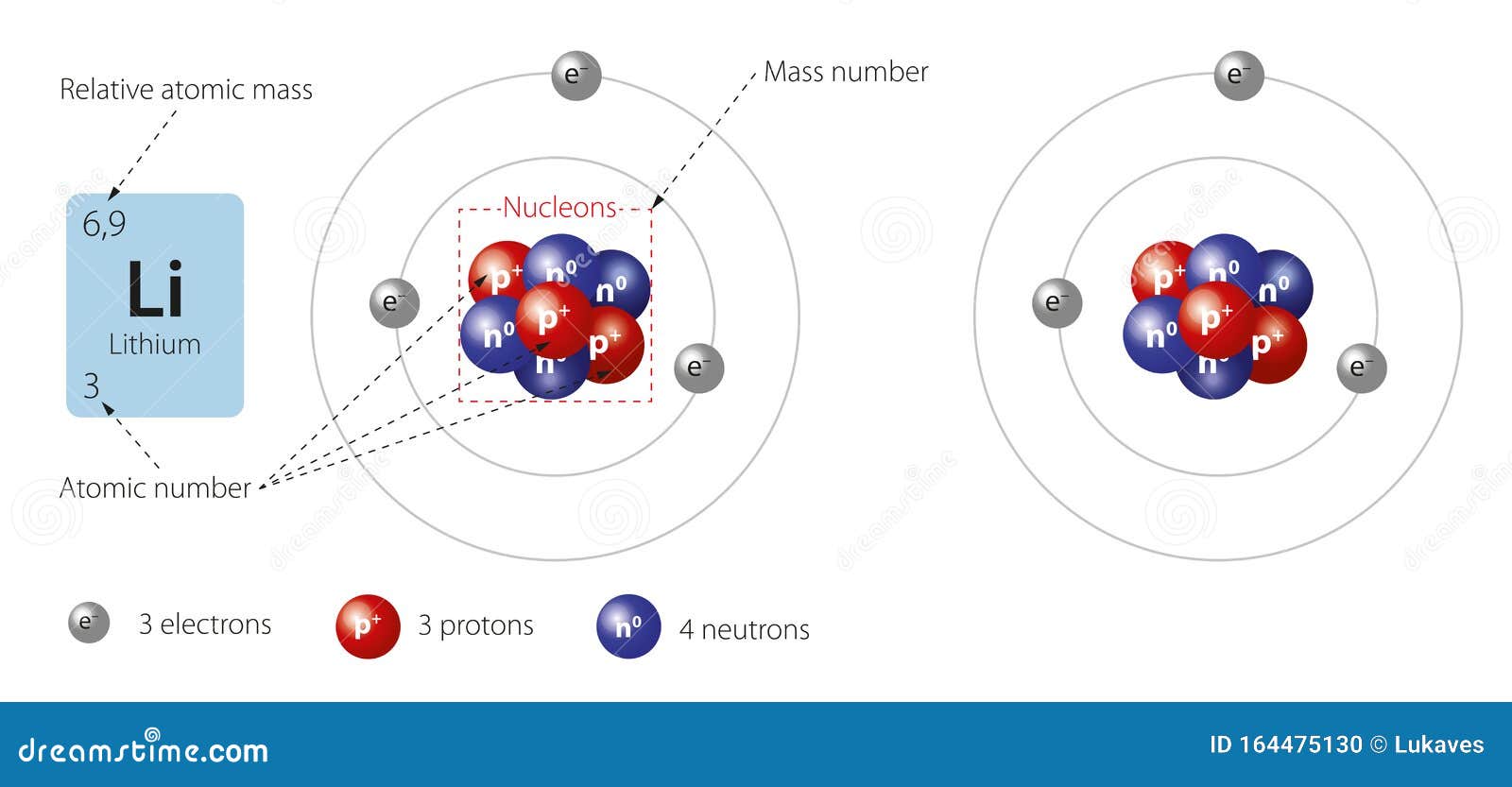
When an atom gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol for example, \(F^-\). Group 17 elements, including fluorine and chlorine, have seven electrons in their outermost shells they tend to fill this shell by gaining an electron from other atoms, making them negatively-charged ions. When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign after the element symbol for example, Na +. As a result of losing a negatively-charged electron, they become positively-charged ions. This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron. In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. 
Their non-reactivity has resulted in their being named the inert gases (or noble gases). As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability they are highly stable as single atoms. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to understand how electrons are distributed in the outer shell of an atom. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.Īn atom may gain or lose electrons to achieve a full valence shell, the most stable electron configuration. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. \):īohr diagrams indicate how many electrons fill each principal shell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
